A taste of our 2017 harvest
-
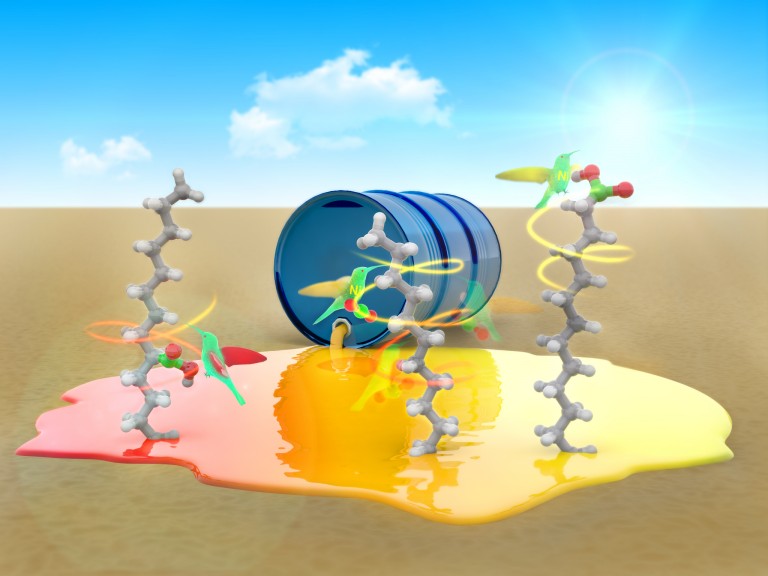
Nickel: a greener route to fatty acids
Chemists designed a nickel catalyst that easily transforms petroleum feedstocks into valuable compounds like fatty acids. The process is environmentally friendly: not only does it work at room temperature and atmospheric pressure, but it also recycles carbon dioxide, contributing to the fight against climate change.
Fatty acids are key in several industrial processes like the manufacture of soaps, plastics –such as nylon– and dyes. Experts estimate that the global market for these compounds could reach $20 billion in the next few years. Classical synthetic methods for obtaining fatty acids often require toxic and hazardous reagents like carbon monoxide and extreme pressures and temperatures. Alternative methods like the derivatization of natural products are less dangerous, but lead to complicated mixtures of products that require laborious purification processes. Last year a team led by Prof. Rubén Martín at ICIQ synthesized a sustainable nickel catalyst that solves both problems. It leads to pure fatty acids from raw hydrocarbons and CO2, which is less toxic than carbon monoxide.
Remote Carboxylation of Halogenated Aliphatic Hydrocarbons with Carbon Dioxide.
F. Juliá-Hernández, T. Moragas, J. Cornella, R. Martin.
Nature, 2017, DOI: 10.1038/nature22316 -
New catalysts mimic human vision
Photoreceptors in vertebrates typically consist of two different, colourless parts: an organic pigment and a protein. When both pieces combine, they create a colourful, light-sensitive molecule –an iminium ion– that triggers vision upon light excitation. Inspired by this mechanism, a team of researchers at ICIQ created a new family of sustainable, environmentally friendly catalysts that can be ‘switched on’ using purple LEDs.
“Despite being a well-known mechanism in biochemistry, the photo-excitation of iminium ions hadn’t been used to make chiral molecules yet,” said Paolo Melchiorre, ICIQ Group Leader and ICREA Professor, who led the study. “Thanks to this novel approach, triggered by visible light, we can obtain products that were impossible to achieve using traditional thermally-activated transformations,” he added. (Read More)
Visible-Light Excitation of Iminium Ions Enables the Enantioselective Catalytic β-Alkylation of Enals.
M. Silvi, C. Verrier, Y.P. Rey, L. Buzzetti, P. Melchiorre.
Nature Chemistry, 2017, DOI: 10.1038/nchem.2748 -
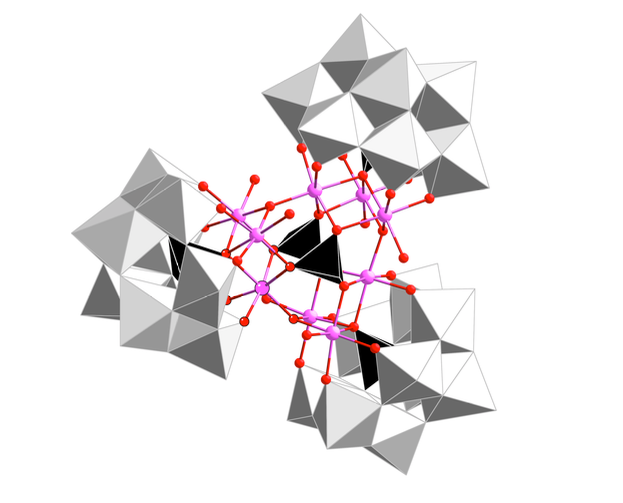
Cobalt and tungsten – together to produce cheaper, cleaner hydrogen
Electrolysis, splitting the water molecule with electricity, is the cleanest way to obtain hydrogen, a clean and renewable fuel. Researchers at ICIQ and URV, led by Prof. José Ramón Galán-Mascarós, designed a new catalyst that reduces the cost of electrolytic hydrogen production.
‘Normally, hydrogen is obtained from using a cheap process called steam reforming. But this is not clean hydrogen, this process uses natural gas and produces carbon dioxide and other contaminants,’ explained Galán-Mascarós. ‘Breaking the water molecule is cleaner, but it’s not easy. We need to develop new cheap, efficient catalysts that allow us to obtain hydrogen at a competitive price,’ he says. To date, the best catalysts are based in iridium oxides, but iridium is a very expensive and scarce precious metal. (Read more)
Polyoxometalate electrocatalysis based on earth-abundant metals for efficient water oxidation in acidic media.
Blasco-Ahicart, J. Soriano-López, J.J. Carbó, J.M. Poblet, J.R. Galán-Mascarós.
Nat. Chem., 2017, DOI: 10.1038/nchem.2874.